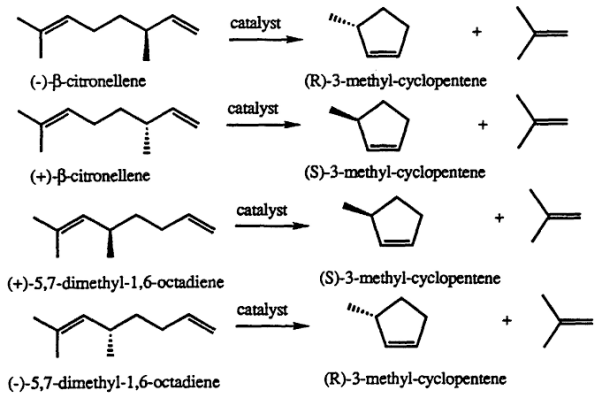
Methylcyclopentene refers to an organic element with two main properties- it is colorless and flammable. It also has a faint odor. The compound is a primary component of napthene, which is a fraction of petroleum. Usually, methylcyclopentene is harvested as a mixture together with cyclohexane. It’s also mainly converted into naphthene, which is a reformer of benzene. 3-methylcyclopentene exists in various forms, but, it is not entirely planar. This implies that it can pucker to alleviate any stress within its structure. Typically, the conversion of this element to benzene is a perfect example of an aromatization reaction. The platinum is then catalyzed to foster the production of gasoline. In this blog post, you will learn more about the chemical and physical reactions of 3- methylcyclopentene with other compounds in various industries.
3- methylcyclopentene has similar characteristics with olefins. However, it has a unique formula. Unlike the olefins, which are also known as straight-chain compounds, 3- methylcyclopentene refers to paraffin bent to form a ring. It can also take a cyclic shape. And just as paraffin, 3- methylcyclopentene is a saturated compound. A few examples of this chemical compound are such as cyclopentane and cyclohexane.

The boiling point of 3- methylcyclopentene and its density are higher than that of various alkanes. Even so, both have carbon. In the real sense, 3- methylcyclopentene is readily available in crude oil. The atom has five to six carbon elements. The multi-ring is also present in all the significant parts of crude oil. While the compound is similar to alkanes when it comes to the physical properties, it has a higher boiling and melting point. It also has a higher density compared to alkanes.
Methylcyclopentene is also an essential element of liquid petroleum products. Its crude oil can be readily converted to gasoline. A single-ring molecular component is heavier than the same volume of alkanes. The chemical and physical structures of methylcyclopentene vary. It also differs in weight and molecular composition. An increase of the polycylic molecules often follows the increase of the weight of this compound.

When it comes to the physical structure of 3-methylcyclopentene, you will be surprised to learn that there is a total of up to 3 cycles identifiable as compounds. But, there is also evidence showing that the number can slightly be higher than the standard expected number. While the investigation of the methylcyclopentene element and its structures and applications is still on-going, people have gained more benefits from this research study over the last ten years. The latest research on the molecular structure of 3-methylcyclopentene indicates that the compound is still undergoing various forms of alterations aimed at improving the physical structure of the compound.
In Closing
In organic chemistry, 3-methylcyclopentene is pretty distinct from other chemical compounds. It is a saturated carbon consisting of hydrogen as well as carbon atoms. The structure has one ring coupled with side chains. Besides, 3-methylcyclopentene has several applications in various sectors. Manufactures of different compounds are still focused on improving the chemical structure of the said elements.